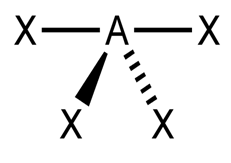
Since H cannot be the central atom, it follows that only C or N can be the central atom. But we know that hydrogen can form only a single bond, this means H cannot be the central atom. 
If you sum all these valence electrons, you will get 10. If you look on the periodic table, you will notice that H has one valence electron, C has 4, and N has 5. Vibrational frequencies were determined for the ClO4- anion and the Li+ClO4- ion pair up to the HF/6-311+G level of theory. Optimized geometries for monodentate, bidentate, and tridentate structures have been determined up to the MP2/6-311+G level of theory. To draw the Lewis structure for HCN, we will first calculate the total number of valence electrons. The anion ClO4- and the ion pair of Li+ClO4- have been studied by ab initio quantum chemical methods. Now, let’s apply the above rules to predict the best Lewis structure for the molecule, 
The first compound is more stable - anion is more stable on a carbon that is. Like charges should not be on adjacent atoms all lone pairs of electrons then, using curved anows to show electron.Negative formal charge should be on the most electronegative atom.How to decide the correct Lewis structure after assigning formal chargesĪfter assigning formal charges, we again apply the following rules to identify the correct Lewis structure: If it is a molecular ion, then the sum of all the formal charges must equal the ionic charge. If it is a neutral molecule, then the sum of all the formal charges must equal zero. It is not a problem because chlorine can keep more than eight electrons in its valence shell (chlorine has 3d orbitals which help to keep more than eight electrons in its last shell. (In ClO 4-lewis structure, there is only one oxygen atom which has three lone pairs) With that, total electrons around chlorine atom is going to be sixteen. Once we add all the formal charges for the atoms in the Lewis structure, we should get a value equal to the actual charge of the molecule or ion. You can convert a lone pair of one oxygen atom which already has three lone pairs to make a bond with chlorine atom. Assign half of the bonding electrons to each atom in the bondĪfter applying the rules outlined above to each atom in the Lewis structure, we will then use the following formula to calculate the formal charge of each atom: How to calculate formal charge.Assign all lone pairs of electrons to the atom on which we find them Its valence diagram is HOCl, with each of the bonds being ordinary covalent bonds, each involving a shared pair of electrons, with one electron coming from each of the atoms at each end of the bond.To determine the formal charge for an atom, we usually follow these rules: When that happens, we usually assign formal charges to the bonded atoms to help determine the correct Lewis structure. Sometimes we can write more than one Lewis structure for a particular ion or molecule. Theoretical investigations support the experimental findings of an intricate network of intermolecular interactions, which is characterised in the studied complex, and also indicate that protonation as well as coordination to the transition metal have important roles in influencing the π-binding properties of the aromatic ring.(© Wiley-VCH Verlag GmbH & Co.Formal charge is the charge we assign to a bonded atom if the bonding electrons were shared equally between the bonded atoms. A high-level ab initio study (RI-MP2/aug-cc-pVTZ level of theory) has been performed to analyse the anion–π binding affinity of the pyridine ring when it is coordinated to a transition metal and also when the other pyridine ring of the 4,4′-bipyridine moiety is protonated. Bader's theory of “atoms in molecules” (AIM) is used to characterise the anion–π and l.p.–π interactions observed in the solid state. If you want the Intro to Chemistry answer, then H2SO4 has a distinct ionic bond between H+ and HSO4. In addition there are π–π and H-bonding interactions in the structure. The noncoordinated carboxylate oxygen is involved in lone-pair (l.p.)–π interaction with the protonated pyridine ring. One of the coordinated perchlorate groups displays anion–π interaction with the coordinated pyridine ring. Each copper atom is also weakly bonded to a perchlorate anion in an axial position. The equatorial plane is formed by the two bidentate picolinate groups in one Cu II, and one picolinate, one monodentate 4,4′-bipyridyl ligand and a water molecule in the other. The structure consists of two copper atoms that have different environments, bridged by a carboxylate group. A Cu II complex of protonated 4,4′-bipyridine (Hbyp) and 2-picolinate (pic),, has been synthesised and characterised by single-crystal X-ray analysis.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
